John good for you for questioning any recommendation you don't understand. What were the test results (as opposed to the recommendations)?
Here is a link to lime/alkalinity and what they are and do and one on water quality. What is your visibility and describe your pond/location. Then we can try to answer the questions. Lime (calcium carbonate) is a buffering agent. I question increasing your ponds ability, through calcium and buffering , to produce phyto-plankton (bloom) and then cutting off its ability to do so by adding dye.
Calcium carbonate is a chemical compound, with chemical formula CaCO3. Calcium carbonate is the active ingredient in agricultural lime.
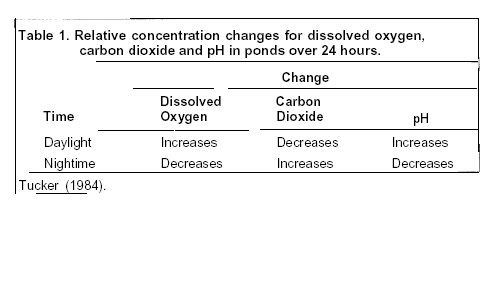
Calcitic limestone (CaCO3) or dolomitic limestone [CaMg(CO3)2], react with the acidity produced by CO2 and dissolves the limestone and form calcium and magnesium bicarbonate salts: CaCO3 + H2O + CO2 = Ca+2 + 2HCO3 or CaMg(CO3)2 + 2H2O + 2CO2 = Ca+2 + Mg+2 + 4HCO3 .The resultant water has increased alkalinity, pH and hardness.
Interactions of pH, Carbon Dioxide,
Alkalinity and Hardness in Fish Ponds
http://srac.tamu.edu/tmppdfs/13452410-46...207d2fa365192a2 A Fish Farmer's Guide to Understanding Water Quality
http://aquanic.org/publicat/state/il-in/as-503.htm